








Preview text:
Scienti c African 26 (2024) e02367
Contents lists available at ScienceDirect Scientific African
journal homepage: www.elsevier.com/locate/sciaf
Effects of plant hormones and genotypes on anther culture
response of safflower (Carthamus tinctorius L.)
Belay Anelay Kassa a,*, Firew Mekbib b, Kebebew Assefa c
a Department of Biology, Bahir Dar University, Bahir Dar, Ethiopia
b School of plant science, Haramaya University, Dire Dawa, Ethiopia
c Debrezeit Agricultural Research Centre, Debrezeit, Ethiopia A R T I C L E I N F O A B S T R A C T Editor: DR B Gyampoh
Safflower (Carthamus tinctorius L.) belongs to the composite family with various uses, from the
stem to the seeds. In vitro flowering and embryo rescue are common techniques for improving Keywords:
safflower hybrid fertility by overcoming breeding constraints. However, anther culture expedites Callus induction
the production of superior types. The aim of the study was to evaluate the effect of plant hor- Genotypes
mones and genotypes on anther culture responses and to develop a suitable protocol. In both Phytohormones Haploids
safflower genotypes, callus, shoot, and root formation were investigated using ten factorial Regeneration
hormone treatments. To promote callus induction and shoot regeneration, variable doses of Safflower breeding
Thidiazuron (TDZ) at 1 mg/l indole butyric acid (IBA) and variable benzylaminopurine (BAP) at
0.5 mg/l NAA were added to the MS medium, respectively. ½ MS media with varying IBA con-
centrations at a fixed 0.5 mg/l NAA were used for root regeneration. Results showed that Turkan
was superior in all aspects of callus induction, and the highest degree of callus formation (46 %)
was observed at 0.5 mg/l TDZ. On the other hand, the local safflower performed better for shoot
and root regeneration. The highest shoot regeneration capacity (20 %) was shown at 2.0 mg/l
BAP, while shoot rooting was reached (21 %) at 1.0 mg/l IBA of culture media. Despite successful
in vitro regeneration, acclimated plantlets did not survive in the glasshouse. In this study, we
demonstrated the higher capability for both shoot and root regeneration of calli produced from
local safflower anthers. More effort is required to improve shoot and root development for both genotypes. Introduction
Safflower (Carthamus tinctorius L.) belongs to the Asteraceae or Composite family, is the only cultivated species in the Carthamus
genus, and was domesticated in the Fertile Crescent over 4000 years ago [50]. Despite its broad range across Asia (including parts of
India) and the Middle East, safflower has a long history of cultivation and is native to Ethiopia, Afghanistan, and other Arab nations
[48]. The cultivated safflower is self-pollinated and has a haploid genome size of about 1.4 GB and 2n = 24 chromosomes [4,12]. It is a
branching herbaceous annual or winter annual oilseed plant with many spines on leaves and bracts [19]. Historically, safflower was
grown for its floral pigments, which were used to make red (carthamin), orange, and yellow (carthamidin) dyes for fabric, foods, and
medicine [60]. Despite this, safflower is used for a variety of purposes, from the stem to the seeds [31]; it is utilized as vegetables, cut * Corresponding author.
E-mail address: belay4ever@gmail.com (B. Anelay Kassa).
https://doi.org/10.1016/j.sciaf.2024.e02367
Received 30 April 2024; Received in revised form 2 August 2024; Accepted 13 September 2024
Available online 19 September 2024
2468-2276/© 2024 The Authors.
Published by Elsevier B.V. This is an open access article under the CC BY license
( http://creativecommons.org/licenses/by/4.0/ ).
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
flowers, fodder, medicinal plants, and textile dyeing [18].
Nowadays, safflower is mainly cultivated for edible oil seed crops in suitable areas of Asia, north-eastern Africa, Australia, the
Americas, and Europe [8]. Its edible oil, which has a 70 % linoleic acid content [5], is used for cooking and salad dressing [26].
Safflower owes its importance to the demand for oil resulting from its high polyunsaturated/saturated fatty acid ratios coupled with
elevated levels of α-tocopherol, which is used to decrease cholesterol levels in the blood and can be used for cardiovascular health [62]
compared to other oilseed crops [24]. Apart from their significance as dietary oils, safflower seeds, oils, and flowers have a plethora of
medicinal uses, such as reducing pain, promoting blood flow, and dispersing blood stasis. Particularly, the natural pigment safflower
yellow (SY) extracted from petals has been widely used in medicine [11] to treat a number of chronic conditions, such as arthritis,
cardiovascular disease, hypertension, and infertility in both sexes [18]. Moreover, safflower flower extracts exhibit anxiolytic prop-
erties comparable to those of diazepam [32]. In general, safflower oil is extensively used in the food, feed, cosmetics, and pharma-
ceutical industries [38] and also has potential in the biofuel industry [44].
In addition to its oil content and medicinal importance, safflower has a wide range of adaptability to a variety of ecophysiological
conditions, making it an ideal crop for drought and salinity tolerance in arid and semi-arid climates around the world [43]. Despite
this, safflower is still a minor crop worldwide. This has been due to its low yield and oil content relative to other oilseed crops and its
susceptibility to diseases and insect pests [23]. Conventional breeding has yielded a few elite cultivars capable of overcoming such
breeding constraints; however, an attempt to produce promising pre-breeding materials from wild safflowers has been unsuccessful
due to significant barriers to sexual cross-ability, variations in basic chromosome numbers, and being mostly self-pollinated [17].
Embryo rescue and anther culture are critical for reducing such barriers as self-incompatibility and sexual incompatibility that might
occur during the breeding process [27]. Embryo rescue is the best in vitro tissue culture technique for enabling the development of a
lethal embryo into a viable plant that carries valuable alleles with agricultural importance from wild species, introduced through
intraspecific, interspecific, or intergeneric crosses [63]. In vitro cultures may also be useful in crosses limited by post-fertilization barriers and embryo abortion.
In this regard, in vitro flowering is a typical event in safflower tissue culture that causes the capitulum to produce fertile pollen and
seeds, as well as novel genetic varieties. This feature could be exploited in recovering wide hybrids [40]. In vitro flowering and embryo
rescue followed by chromosomal doubling with colchicine are common techniques for improving safflower hybrid fertility, even
though anther culture speeds up the generation of superior types [59]. Recently, anther culture has been suggested as a fast way to
generate haploid and totally homozygous lines [30], which shortens the time required to produce pure lines with commercially viable
varieties [37]. Pure lines offer substantial benefits over conventional techniques for introducing them into agriculture, since dihaploid
plants with doubled chromosomes can achieve full homozygosity at every locus in a single generation by seed multiplication [61]. For
this reason, anther culture works well as a substitute for the conventional method of developing inbred lines in safflower, which
involves several inbreeding cycles.
Anther culture is essential for quickly producing new cultivars of safflower; however, in vitro safflower shoots are prone to
hyperhydration, and inadequate rooting is a major barrier to both safflower transformation [7] and also to safflower anther culture
[45]. Associated constraints still prevent the realization of the full potential of safflower haploid regeneration. A number of factors,
such as genotype, growth conditions of the donor plant, cold pre-treatment, and growth hormones, also influence the culture efficiency
of safflower [37]. The success of the anther culture technique cannot be proven with respect to genotypes and hormonal treatments,
and utilizing this method for safflower breeding would require fine-tuning the anther culture procedure while taking into account the
limitations. Apart from that, there hasn’t been much research done on safflower anther culture up to this point, not just in Ethiopia but
in other places as well. Therefore, the aim of the study was to evaluate the effect of plant hormones and genotypes on the anther culture
responses and further develop a suitable and reliable safflower anther culture technique that can be used for in vitro germplasm improvement. Materials and methods Plant material
The seed materials of two safflower genotypes (Local and Turkan) were obtained from the Highland oil crops research department
of the Holleta Agricultural Research Centre. The local safflower variety is of Ethiopian origin, while Turkan is the exotic variety. Both
safflower genotypes show unique variations in seed morphology and post-germination characteristics. The Turkan safflower seed is
large, smooth, and has no spines on the leaves, in contrast to the local safflower seed, which is small, rough, and has many spines on its
leaves. The experiment was conducted at the Debre Zeit Agricultural Research Centre’s (DZARC) tissue culture laboratory. The donor
plants were cultivated and maintained in the glasshouse at DZARC until flowering started.
Culture media and treatments
A full-strength MS [36] basal medium with different concentrations and combinations of plant growth regulators was prepared. A
factorial combination of two safflower genotypes (local and Turkan) and five media with different concentrations of TDZ and 1.0 mg/l
IBA were used for callus induction. This hormonal combination can be the most effective treatment for eliminating the secretion of
phenolic substances. The MS medium was prepped as follows: 1) 0 mg/l TDZ + 1.0 mg/l IBA (T1); 2) 0.25 mg/l TDZ + 1.0 mg/l IBA
(T2); 3) 0.5 mg/l TDZ +1.0 mg/l IBA (T3); 4) 0.75 mg/l TDZ +1.0 mg/l IBA (T4); and 5) 1.0 mg/l TDZ +1.0 mg/l IBA (T5) for callus
induction. All the media were supplemented with mineral salts, vitamins, 3 % sucrose, and 0.8 % agar. The pH of the medium was 2
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
adjusted to 5.6 with 1 N HCl and 1 N NaOH before adding agar and autoclaving. Also, every piece of equipment, including glasses and
utensils utilized for each tissue culture activity, was autoclaved, and the workspace was thoroughly cleaned. Plant material of
immature capitula of all genotypes was gathered in the morning before 10 a.m., when the plants were in the late uni-nucleate stage. Then
1) The collected capitula were wrapped in aluminum foil and kept in a refrigerator at 5 ◦C for 24 h.
2) Prior to culture, the juvenile capitula were sterilized by being rinsed in 70 % ethanol for 1 min, in 25 % hypochlorite bleach for 10
min, and finally washed three times with sterilized distilled water.
To ascertain the pollen development stage, the anthers were dissected, inspected using a 1 % acetocarmine stain, and determined
by light microscopy. The uninucleate stage of anthers was chosen for culture because microspores at this stage are advantageous for
many genotypes and species in anther cultures. Typically, florets are white at the early-late uninucleate stage; florets become yellow as
they mature, and the colour of the floret was also employed as an index to select the anthers with early-late uninucleate microspores.
Anther culture for callus induction
Anthers were aseptically dissected using sterilized forceps and needles in a UV-sterilised laminar flow hood. Ten anthers were then
inoculated onto 60 × 15 mm-diameter plastic Petri dishes containing 10 ml of MS medium with five hormonal concentration treat-
ments. The petri dish plates containing cultivated anthers were sealed with parafilm and kept in complete darkness at a temperature of
25 ± 1 ◦C. Petri dish plates were inspected once a week, and information on callus induction was noted and recorded, including the
number of responding anthers, callus formation percentage, days of callus formation, callus weight, callus nature, and callus colour. Shoot regeneration
After six to seven weeks of anther inoculation, the produced calli were aseptically removed and transferred to a sterile culture vessel
medium. The calli were broken up into tiny bits and placed into a small magenta that contained 30 ml of shoot regeneration media.
Sub-culturing in the MS media was done with different BAP (0, 1, 1.5, 2, and 2.5 mg/l) and NAA (0.5 mg/l) doses. Using a 16/8-hour
photoperiod (days/night), the magentas were incubated at 25◦ ±1 ◦C. The number of calluses that responded, the percentage of
regeneration, the number of calluses with a shoot in each jar, the number of shoots per total callus of each treatment, the number of
regenerated shoots per callus, and other information related to shoot regeneration were all recorded.
Root induction and acclimatization
After the sub-culture calli produced proliferated and differentiated shoots with a length of roughly 2–3 cm, the shoots were
aseptically removed from the cultivated magenta, separated from each other, and then cultured again on root induction medium. Sub-
culture was done on half-strength MS media containing different (0, 0.5, 1.0, 1.5, and 2 mg/l) concentrations of IBA with 0.5 mg/l NAA
and 1 % sucrose in freshly prepared root induction medium and incubated under continuous light. Statistics on root regeneration,
including the number of responsive shoots, the number of roots per plant, and the percentage of roots formed during root induction,
were recorded. After that, plantlets with a significant amount of roots were taken out, and the associated media was gently cleaned out
with running tap water. Then, the plantlets were transplanted into pots containing a mixture of garden soil, sand, and cow dung in the
ratio of 1:2:1. Immediately after transplanting, the plants along with pots were covered with polythene bags to prevent desiccation. To
reduce sudden shock, the pots were kept in the growth room for 7–15 days in a controlled environment. Data analysis
All experiments and treatment combinations were laid out in a completely randomized design (CRD) with three replications. This
design is vital for minimizing systematic bias and supporting the validity of experimental conclusions. The data for each of the callus
induction, shoot regeneration, and root induction parameters were recorded. The analysis of variance was conducted using SAS
computer software version 9.0, and the significance of differences between the pair of means was evaluated at the 5 % level of sig-
nificance by Duncan’s multiple range test (DMRT) and expressed as the mean ± standard error. Result and discussion
Genotypic effects on callus induction
In vitro androgenic responses are highly genotype-dependent, and the concentration of exogenous hormones would affect the
developmental pathway of pollen and callus induction frequency [3]. Callus induction is one of the steps needed for haploid plant
regeneration, and callus initiation growth regulators are essential for the haploid plants to develop [20]. The mean values of genotypes
for the parameters indicated in the Table were found to be statistically significant. For all treatments, the Turkan genotype showed
significantly better callus induction responses than the local genotype for all parameters considered. This study demonstrated that the
two genotypes varied widely in their callus induction frequency and responded differently at various concentrations of TDZ. On
average, over both safflower genotypes, TDZ concentration showed substantial variations for the number of anthers with callus in-
duction, percent of callus formation, number of calli/petri-dish, and callus weight. The Turkan genotype anther displayed a greater 3
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
callus formation rate (69.33) and induction frequency (46.20 %) than the local genotype callus formation rate (48.66) and induction
frequency (32.40 %), which was cultivated at 0.5 mg/l TDZ + 1 mg/l IBA.
The current investigation showed that the two genotypes responded differently to different TDZ concentrations and varied greatly
in their callus induction frequency (Fig. 1a and b). Genotypic effects have been reported for the Turkish and Iranian safflower cultivars,
where the Turkish cultivar (Dincer) had a better callus induction response than the Iranian cultivar (Sina) cotyledon explants cultured
on MS media supplemented with 0.5 mg/l TDZ + 0.25 mg/l IBA [34]. Our observation is supported by the finding of Silva and
Ratnayake [55], where androgenic responses varied among cultured anthers of different japonica rice. The differences in callus in-
duction ability between the two safflower genotypes subjected to different growth regulator concentrations suggest that genotypes
may have different endogenous hormone levels, as noted by Obert et al. [42] with callus induction of anther culture variations among
flax genotypes at different growth regulator concentrations. This also agreed with, where callogenesis responses varied depending on
the cultivar and medium composition interactions.
In the present study, reduced frequencies of callus induction were noted at both lower and higher concentrations of TDZ. The least
amount of callus formation was found in MS medium supplemented with 1.0 mg/l TDZ + 1.0 mg/l IBA and 0.0 mg/l TDZ + 1.0 mg/l
IBA, at 13.96 % and 20.20 %, respectively. This investigation exhibited that both the high and low concentrations of TDZ influence
callusing performance; however, calli induction rates were reduced with further increments of TDZ. This outcome agrees with the
finding of Srivastava et al. [57] that Brassica oleracea’s callus induction decreased in an enriched medium at both lower and higher TDZ
concentrations. Additionally, Duan et al. [16] found that callus development was not supported by either high or low TDZ concen-
trations, with the highest rate of callus induction (67.78 %) observed for Paeonia lactiflora leaf explants cultured at 1.0 mg/l TDZ + 1.0
mg/l 2,4-D. All parameters were found to be highly significant for genotype and TDZ concentration interactions. The Turkan genotype
grown on MS medium containing 0.5 mg/l TDZ + 1.0 mg/l IBA produced the highest number of callus-induced anthers (69.33), while
the local genotype grown on MS medium containing 0.0 mg/l TDZ + 1.0 mg/l IBA produced the lowest number of anthers (21.00).
The highest callus formation frequency (46.20 %) was observed in the Turkan genotype cultivated on MS medium containing 0.5
mg/l TDZ and 1.0 mg/l IBA; however, the local genotype cultured on the medium without TDZ showed the lowest callus formation
frequency (13.96 %). This is consistent with the results of Rayan and Awad [46], where the higher callus induction effects of TDZ
combined with IBA were observed in apple anther culture and Iranian cannabis cotyledon cultures by Movahedi et al. [35]. Similarly,
Zhou et al. [64] confirmed the efficiency of the TDZ and IBA combination for callus induction, wherein callus growth was greatest and
the induction rate was highest (54 %) in pear leaves cultured at 3.0 mg/l TDZ + 4.0 mg/l IBA, despite the hormone concentration being
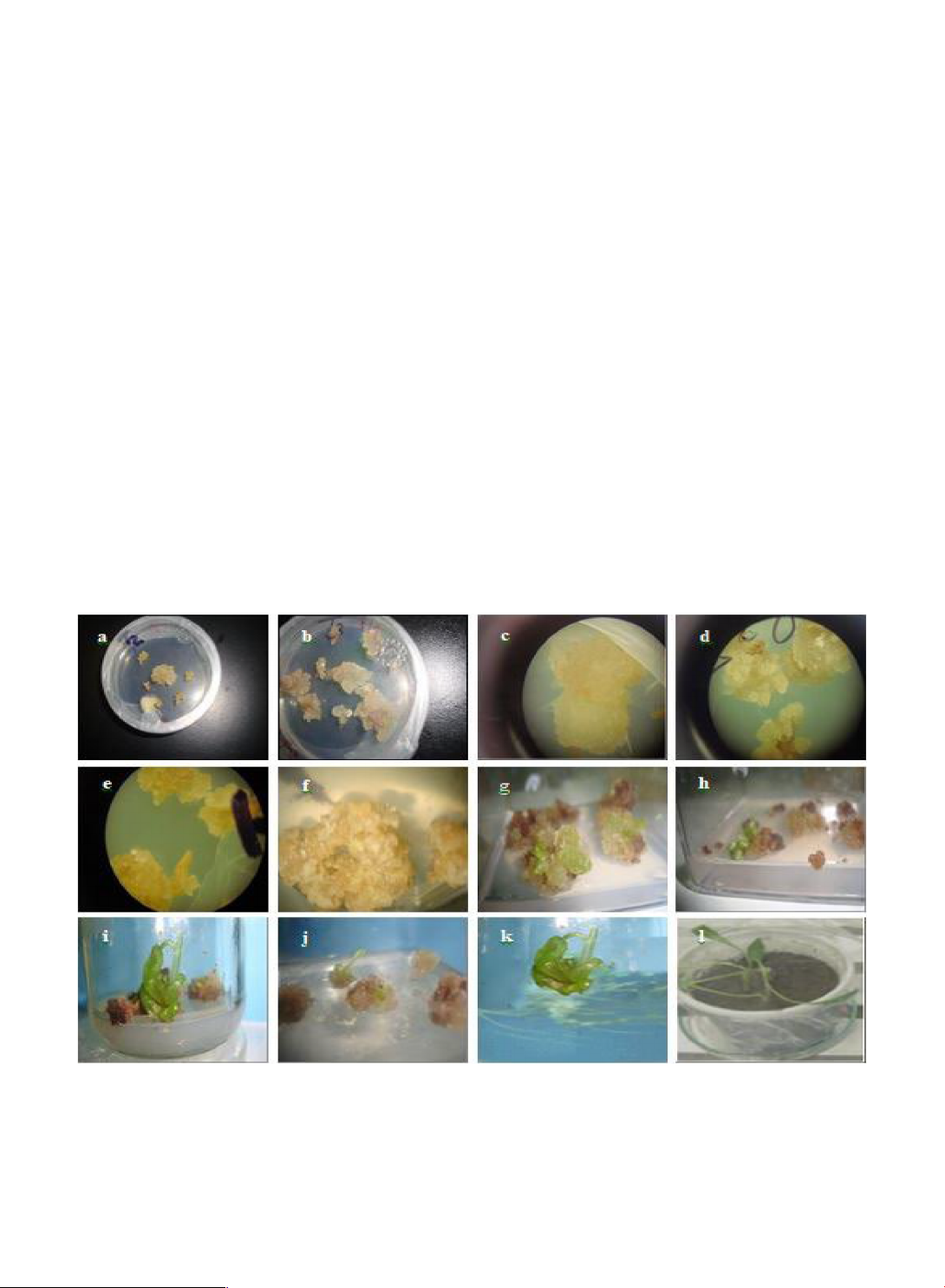
Fig. 1. Effect of genotypes and media hormone composition on the callusing responses, shoot regeneration, root induction and acclimatization.
A) Local safflower anthers cultured at 0.25 mg/l TDZ and 1.0 mg/l IBA, B) Turkan safflower anthers cultured at 0.5 mg/l TDZ and 1.0 mg/l IBA, C)
Compact and organized callus in the local safflower genotype, D) Friable and non-organized callus in Turkan safflower genotype, E) Pale yellow
color callus in Turkan safflower genotype, F) Highly proliferated and irregular-shaped calluses in the Turkan safflower, G) Green globular patches
and leaf-like features on the local safflower genotype callus; and H) Shoot-like buds are present in the local safflower callus, I) Regenerated shoots
from the local safflower genotype, anther-derived callus, J) Regenerated shoots from the anther-derived callus of the Turkan safflower, K) Root
regenerated shoot in local safflower and L) Acclimatization of plantlets in the growth room. 4
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
high. In a similar vein, the effectiveness of TDZ in inducing callus formation was observed in Hibiscus syriacus leaf explants grown on
MS media at 0.5 and 1.0 mg/l of TDZ concentrations [52], and the addition of 0.2 mg/l or 0.5 mg/l of TDZ in the medium led to
improved callus formation in blueberry [10]. Even though the hormone concentrations varied, the maximum callus induction rate
(92.0 %) was observed in the okra anthers grown at 0.1 mg/l TDZ + 2.0 mg/l IAA treatment [20]. Furthermore, TDZ and IBA’s
cytokinin/auxin combination was found to have substantial callus induction efficiency; its efficiency was significantly lower than that
of TDZ and the 2, 4-D combination for runner tips of strawberry explants [1].
Genotypes varied significantly in the number of days needed for callus initiation; in the anther, callus initiation typically started
14–28 days after incubation and ended 35–42 days later. The Turkan genotype, grown on MS medium containing 0.5 mg/l TDZ,
exhibited the quickest callus emergence (27.06 days), while the local genotype, grown on MS medium containing 0.0 mg/l TDZ,
showed the most delayed callus start (39.66 days). Similarly, in the anther cultures of rice genotypes, the number of days needed for
callus induction varied and ranged from 32 to 55 days [14,53]. The genotype and TDZ concentration interaction were found to have a
significant effect on the days required for callus initiation. Similar results were found by Alam et al. [2] on brassica anther culture,
where different days were taken for callus initiation and completion. Furthermore, following six weeks of inoculation, two genotypes
showed statistically significant differences in the number of calli per petri dish (Table 1). Remarkably, we observed that the two
safflower genotypes differed in callus morphology, such as being compact and friable (Fig. 1c and d). This indicates that callus in-
duction and morphology vary with the genotype of plants. Nakas et al. [37] corroborate this finding, showing that only two of the
twelve safflower genotypes in the anther culture, cultivars Balcı and Linas, developed regenerable calli; the other genotypes did not
produce compact and green calli, which are more likely to initiate shoots. Our result showed that the in vitro androgenic and
morphogenic responses are highly genotype-dependent. Aside from genotype, this could be the result of TDZ concentration. The
potential of TDZ in the formation of various callus morphologies was investigated by Seo et al. [52], yielding friable, compact, and
yellowish calli from all Hibiscus syriacus leaf explants cultured on MS medium containing 0.01 mg/l TDZ. On the contrary, Ribeiro et al.
[47] found that the use of TDZ only caused the formation of compact calluses in Hovenia dulcis explant culture. Furthermore, callus
formation might be determined by biochemical parameters of the medium, such as the carbon and nitrogen sources, in addition to the
concentration and genotype type of the plant.
Shoot regeneration responses
Shoot regeneration from the anther-derived callus is a critical phase of the androgenetic process in haploid plant production. The
two safflower genotypes differed significantly in terms of shoot initiation from the anther-derived calli, and neither genotype showed a
correlation between callus induction and shoot regeneration. The local genotype had the largest number of calli (15.93) and a higher
mean frequency (10.48 %), while the Turkan genotype had the lowest number (8.86) and the lowest frequency (5.83 %) of shoot-
regenerating calli. Across all BAP concentrations, the local genotype was highest in the mean number of shoots/responding callus
(3.05) and shoots/total callus (0.39), and the Turkan genotype had the lowest mean (2.72) and (0.19) for each respective parameter. Table 1
Effects of different concentrations of TDZ along with 1 mg/L of IBA for callus induction from anthers of two safflower genotypes (the number of
anthers cultured for each of the treatment combinations were 450). Treatment NAR PCF DCF NC/PD CW(g) Genotype TDZ(mg/L) Local 0 21.00f 13.96f 39.66a 1.40f 0.90f 0.25 58.00b 38.60b 32.20de 3.86b 0.76g 0.5 48.66c 32.40c 33.13cd 3.24c 1.09e 0.75 40.33d 26.83d 36.40b 2.68d 1.22d 1.0 30.33e 20.00e 35.00bc 2.02e 1.53b Turkan 0 27.33e 18.20e 30.80ef 1.82e 0.93f 0.25 54.66b 36.43b 29.86f 3.64b 0.93f 0.5 69.33a 46.20a 27.06g 4.62a 1.34c 0.75 49.66c 33.10c 29.86f 3.31c 1.40c 1.0 36.00d 23.96d 29.86f 2.40d 1.85a
Means of Genotypes (Over all TDZ concentration treatments) Local 39.66b 26.36b 35.28a 2.64b 1.10b Turkan 47.40a 31.58a 29.49b 3.16a 1.30a
Means of TDZ concentration treatments (Over both safflower genotypes) 0 24.16d 16.08d 35.23a 1.60d 0.92d 0.25 56.33a 37.52a 31.03cd 3.75a 0.84d 0.5 59.00a 39.30a 30.10d 3.93a 1.22c 0.75 45.00b 29.96b 33.13b 2.99b 1.31b 1.0 33.16c 21.98c 32.43bc 2.20c 1.69a OM±SEM 43.53 ± 7.233 28.97 ± 3.327 32.38 ± 1.372 2.9 ± 0.032 1.20±0.004 CV (%) 6.18 6.29 3.62 6.18 5.05
* Means in the same column and within the same treatment category followed by different superscript letters are significantly different at P ≤ 0.05 as
judged by DMRT, OM = overall mean, SEM = Standard Error of the Mean and CV = Coefficient of Variation, NAR = number of anthers responding,
PCF = Percept of callus formation, DCF = Days to callus formation, NC/PD = number of calli per petri dish and CW = callus weight in gram. 5
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
Similar results were also observed for shoot length, where the local safflower genotype had the maximum shoot length (2.01 cm) and
the Turkan safflower genotype had the minimum shoot length (1.73 cm) (Table 2). Such an outcome demonstrates that the two
safflower genotypes differed in their shoot regeneration efficacy, and the occurrence of genetic variations is very common in vitro
cultures, especially during indirect organogenesis from callus. Based on the observed result, we concluded that exogenous regulators
determine the successful formation of shoots, while endogenous hormone levels define the response in vitro.
This observation is correlated with the result of Sujatha and Dutta [58], where both direct and callus-mediated shoot regeneration
differences were observed among safflower genotypes. Similarly, Sakina et al. [49] reported that green plant regeneration was
significantly varied in the anther cultures of different rice genotypes. The variation in shoot regeneration between the genotypes may
be associated with callus quality. The Turkan genotype may have had poorer shoot regeneration due to its friable calli, whereas the
majority of the calli in the local genotype were more compact, which may have improved shoot regeneration. This observation is in
agreement with Nakas et al. [37], who found that the anther-derived calli of the safflower genotypes did not develop into compact and
green calli that are less likely to start shoots. This result is also in line with Mosoh et al. [33], who found that more compact and green
calli promote shoot development and have the highest number of shoots on their surface.
Additionally, there were substantial differences in shoot regeneration associated with the BAP concentration. When considering
shoot regeneration frequency among other parameters, the highest values of shoot regeneration frequency (16.99 %) were seen at 2
mg/l BAP + 0.5 mg/l NAA, followed in that diminishing order of 1.5 mg/l of BAP (9.86 %), 2.5 mg/l of BAP (9.10 %), and 1.0 mg/l of
BAP (4.38 %), respectively. On the other hand, the lowest values for all significant metrics were observed on the MS medium without
BAP enrichment. This difference suggests that the cytokinin level in the medium influences shoot formation, as noted by Javad et al.
[21], where different BAP concentrations increased shoot regeneration from the cotyledon-derived callus in both Turkish and Iranian
safflower cultivars (Dincer and Sina), but the Dincer cultivar exhibited the highest percentage of shoot regeneration on a medium
containing 2 mg/l BAP and 0.1 mg/l NAA. Likewise, Chung and Ouyang [13] found that different strawberry cultivars have specific
hormonal requirements for shoot regeneration and show a variable regeneration rate.
Furthermore, the link between BAP concentrations and genotypes was found to have a significant impact on shoot regeneration
parameters. Among the different BAP concentrations and genotypes, the local genotype performed better with BAP at 2 mg/l. Some of
the characteristics taken into consideration in shoot regeneration are the average shoot length (2.85 cm), percent of shoot formation
(19.95 %), and calli with shoot development (30.33 %) (Table 2). Similarly, the highest percentage of shoot regeneration was observed
from both cotyledon and anther-derived safflower explants on the MS medium containing 2 mg/l BAP and 0.5 mg/l NAA in Dincer
[34]. This was also supported by Kumar et al. [25], who found that Marigold anthers showed the best shoot regeneration on the MS
medium supplemented at 2.0 mg/l BAP + 0.5 mg/l NAA, and by Nazir et al. [39], where BAP 2 mg/l + NAA 0.5 mg/l showed excellent
shoot regeneration percentages in Valeriana jatamansi Jones.
Even if shoot regeneration differences were noticed between genotypes, the overall callus-derived shoot regeneration potential of
the two safflower genotypes was very poor. While both safflower genotypes exhibited inadequate differentiation of embryoid and Table 2
Effects of different concentrations of BAP along with 0.5 mg/l of NAA for shoot regeneration from anther derived callus of the two safflower genotypes
(the number of callus were 152 for each of the treatment). Treatments NCR PSF NCS/J NS/TC S/RC SL (cm) Genotype BAP (mg/L) 0 1.33e 0.88e 0.04f 0.01g 0.78e 0.58f 1.0 7.33cd 4.82cd 0.19de 0.12f 2.70d 1.76e Local 1.5 21.33b 14.04b 0.55b 0.45c 3.26c 2.53b 2.0 30.33a 19.95a 0.79a 0.78a 3.91b 2.85a 2.5 19.33b 12.72b 0.49c 0.58b 4.62a 2.34bc 0 0.00e 0.00g 0.00f 0.00g 0.00f 0.00g 1.0 6.00d 3.94d 0.16e 0.13ef 3.14c 1.93de Turkan 1.5 8.66c 5.70c 0.23d 0.17e 3.07cd 2.40bc 2.0 21.33b 14.04b 0.56b 0.46c 3.29c 2.22cd 2.5 8.33c 5.48c 0.22d 0.22d 4.13b 2.09cd
Means of Genotypes (Over all BAP concentration treatments) Local 15.93a 10.48a 0.42a 0.39a 3.05a 2.01a Turkan 8.86b 5.83b 0.23b 0.19b 2.72b 1.73b
Means of BAP concentration treatments (Over both safflower genotypes) 0 0.66d 0.44d 0.02d 0.01e 0.38d 0.28d 1.0 6.66c 4.38c 0.18c 0.12d 2.92c 1.84c 1.5 15.00b 9.86b 0.39b 0.31c 3.17c 2.46a 2.0 25.83a 16.99a 0.67a 0.62a 3.60b 2.53a 2.5 13.83b 9.10b 0.36b 0.40b 4.38a 2.22b OM±SEM 12.39±1.66 8.16±0.72 0.32±0.00 0.29±0.00 2.88±0.06 1.87±0.03 CV (%) 10.41 10.42 10.34 9.45 8.51 8.96
* Means in the same column and within the same treatment category followed by different letters are significantly different at P ≤ 0.05 as judged by
DMRT or t-test. OM = overall mean, SEM= Standard Error of the Mean, CV=Coefficient of Variation, NCR = number of callus responding, PSF=
percent of shoot formation, NCS/J = number of calli with shoot per Jar, NS/TC = number of shoot per Total Calli, S/RC = Shoot per Responding Calli and SL= Shoot length. 6
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
plantlet, the local safflower outperformed the Turkan in terms of regeneration and shoot performance (Fig. 1i and j). The results are in
agreement with those of Lijiao and Meili [28] and Nikam and Shitole [41], who reported that safflower shoot regeneration was fairly
harder than that of other congeneric plants. The shoot regeneration problem from anther culture has been previously reported in the
production of haploids in many crops, including rapeseed [9], peppers [51], and rice due to albino shoots [29]. Another noteworthy
observation of this study was that even safflower tissues exhibit a great propensity for multiple shoots from a few anthers derived from
the callus, and a higher number of shoots is invariably associated with hyperhidricity and shoots without distinct nodes. Those are the
most infuriating problems in safflower tissue culture. Apart from the genotype of the crop, the aforementioned problems could be TDZ
side effects, as excessive TDZ exposure has been linked to hyperhidricity, abnormal shoot growth, and difficulty in root generation
[15]. As a result, all safflower genotypes had a low incidence of shoot regeneration, which may be connected to the plant’s ability to
regenerate. Genotype is one of the most important factors that could affect how well the callus generated from an anther regenerates.
Root regeneration and acclimatization
The formation of a functional root system is one of the most important milestones in the development of plant tissue culture
techniques. However, rooting and acclimatization of in vitro plants have been a bottleneck for successful propagation [6]. The
establishment of a functional root system is a vital stage in a plantlet’s ability to acquire water and mineral nutrients as well as adapt to
stress under post-vitro conditions [22]. The local safflower genotype exhibited a variable root regeneration response for its cultured
shoots on the half MS medium supplemented with different IBA concentrations; however, the Turkan safflower genotype did not
exhibit any root-regenerating shoots. With regard to all IBA concentrations, the local safflower genotype showed the highest mean
number of regenerating shoots (11.20), the largest mean number of roots (2.69), and the greatest mean root formation response (18.66
%). Fortunately, the Turkan safflower genotype did not generate roots in its shoots, and the number of root-forming shoots varied
significantly between the safflower genotypes. This indicated that genotype significantly affects root regeneration efficiency since the
in vitro rooting process is governed by diverse genetic factors that have strong effects on organogenesis.
The problem of root development from shoots produced through safflower tissue culture was similarly reported by Sujatha and
Dutta [58]. Besides, IBA concentrations varied considerably for root-initiation; the highest mean shoots with root growth response
(10.50) and the largest mean root formation response (17.49 %) were observed at 1 mg/l IBA- containing MS medium. The MS medium
enriched with 1.5 mg/l IBA showed the highest mean number of roots per shoot (1.92). However, the cultured shoots did not form any
roots in the medium lacking IBA (Table 3). For the local genotype, the culture medium with 1.5 mg/l IBA produced the highest mean
number of roots per shoot (3.85), while the MS medium with 1 mg/l IBA showed the best root establishing (21.00) and greatest mean
root formation frequency (35.00 %). Over all, the best root formation response was found on the MS medium supplemented with 1
mg/l IBA + 0.5 mg/l NAA for the local genotype. Similar results were obtained by Alam et al. [2] in Brassica species and found the
highest root regeneration response at 1.0 mg/l IBA along with 0.5 mg/l NAA, as well as a higher root formation frequency observed at
the IBA and NAA hormonal combination [6]. Table 3
Effects of different concentrations of IBA along with 0.5 mg/l of NAA for root regeneration from anther derived shoots of the two safflower genotypes
(the number of shoots cultured for each of the treatment combinations were 60 and 30 in local and Turkan, respectively). Treatments NSR NR/S PRF Genotype IBA (mg/l) Local 0 0.00e 0.00e 0.00e 0.5 12.00c 2.76d 19.99c 1.0 21.00a 3.53b 35.00a 1.5 15.00b 3.85a 24.99b 2.0 8.00d 3.32c 13.33d Turkan 0 0.00e 0.00e 0.00e 0.5 0.00e 0.00e 0.00e 1.0 0.00e 0.00e 0.00e 1.5 0.00e 0.00e 0.00e 2.0 0.00e 0.00e 0.00e
Means of Genotypes (Over all IBA concentration treatments) Local 11.20a 2.69a 18.66a Turkan 0.00b 0.00b 0.00b
Means of IBA treatments (Over both safflower genotypes) 0 0.00e 0.00e 0.00e 0.5 6.00c 1.38d 9.99c 1.0 10.50a 1.76b 17.49a 1.5 7.50b 1.92a 12.49b 2.0 4.00d 1.66c 6.66d OM±SEM 5.60±0.40 1.34±0.00 9.33±1.11 CV %) 11.29 2.95 11.29
*Means in the same column and within the same treatment category followed by different letters are significantly different at P ≤ 0.05 as judged by
DMRT or t-test, OM = overall mean, SEM = Standard Error of the Mean and CV = Coefficient of Variation, NSR = number of Shoots Responding, NR/S
= number of Root per Shoot, PRF = Percent of Root Formation. 7
B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
Notably, neither the Turkan genotype planted shoots nor the majority of the local genotype planted branches had a response. The
planted shoots exhibited callus formation at the cut end, drying of the shoot tip, and swelling of the shoot base, all of which further
hampered root growth and caused the shoots to become vitrified. Similar problems were observed with in vitro-rooted safflower shoots,
which decreased the overall effectiveness of whole plant regeneration [34]. In this investigation, the regenerated roots functioned
badly in the local safflower; however, there were no root regenerations in the Turkan safflower. This outcome was in agreement with
Sujatha and Dutta [58], who demonstrated a safflower root regeneration study using growth regulator-free or IBA-enriched MS media
and revealed that rhizogenesis and root formation frequency were not as high as anticipated. This problem was further validated by
Nakas et al. [37], who noted that the safflower anther shoots had inadequate root formation and root appearance subsequent to the
shoots’ death from hyperhydration. Although root formation is one of the most essential stages in plant tissue culture, it directly affects
greenhouse survival and acclimatization success [54].
The failure of any plantlets to survive after acclimatization is another important discovery made in this study. The survival rate of
those plantlets in plastic pots was low, and most of the plantlets were unable to acclimate after two weeks in the growth chamber. Of
the plantlets acclimatized for two weeks in the growth room, only four (33.33 %) were transferred into the glasshouse; however, none
of the plantlets could survive even a week in the glasshouse. In vitro regenerated shoots must efficiently establish roots in order to
acclimate, yet safflower rooting and physiological changes continued to be problematic. Even though the formation of strong and
robust roots is necessary for efficient acclimatization in ex vitro conditions and subsequent field transfer [6], the plantlets could not
survive in the glasshouse for a week. This is due to insufficient root formation and weak root performance, which enhance the survival
rate of the plantlets. Similar problems were reported by Sri Shilpa et al. [56], with limited in vitro root development and in vitro-induced
root performance, in which these roots often die after being transplanted to soil from tissue culture media containing cotyledon-formed safflower roots. Conclusion
Anther culture is a biotechnology tool that develops pure lines in a short time, thereby shortening the breeding period. In this study,
the responses of two safflower genotypes to anther culture were different depending on the plant growth regulators for each stage of
the experiment. The effectiveness of TDZ in inducing callus tissue was demonstrated in both safflower genotypes, despite notable
differences observed between genotypes, with Turkan exhibiting superior performance across all callus induction metrics. Growth
regulators should always be considered in a range of concentrations, and the largest and most callus-inducing anthers were observed
on MS medium containing 0.5 mg/l TDZ and 1.0 mg/l IBA in the Turkan safflower. In shoot regeneration, the local safflower performed
better in vitro shoot regeneration than the Turkan genotype, despite both genotypes showing limited potential. The BAP 2 mg/l and
NAA 0.5 mg/l-supplemented MS medium showed the greatest shoot regeneration efficiency in the local safflower calli. The Turkan
safflower genotype, on the other hand, had both low shoot regeneration potential and underperforming emerging shoots. Shoot
elongation was very important for rooting; thereby, root induction potential was poor in the local genotype, and no response was
observed in the Turkan safflower genotype. The root induction MS medium at 1 mg/l IBA and 0.5 mg/l NAA showed a superior root
development response in the local safflower compared to the other phytohormone concentrations. Furthermore, the plantlet survival
rate during acclimatization in the growth room was extremely low, and not a single plantlet acclimatized in the glasshouse. In general,
in vitro shoot and root regeneration from anthers-derived callus was very low in both safflower genotypes, and the most typical issues
observed in this study were inadequate roots and hyperhydricity of the shoots. Further research will be necessary to optimize growth
regulator combinations and genotype influences on shoot and root regeneration, as well as the survival rate of rooted shoots, in order
to develop useful in vitro anther culture protocols that will aid in a practical safflower breeding program.
CRediT authorship contribution statement
The journal requests that authors provide an author statement file detailing their specific contributions to the paper using the
appropriate CRediT roles. Regarding our publication, the research team members are listed in the authors’ contribution CRediT who
have significantly contributed to the conception, design, collection, processing, and interpretation of the article as: Belay Anelay
Kassa: Conceptualization (lead); Methodology, Software, Data Collection, organization to data processing and analysis (lead); writing
and editing the original paper draft. Firew Mekbib: Conceptualization (supporting); Supervision, writing–review and editing.
Kebebew Assefa: Resources, Supervision, Validation, Software, formal analysis (lead); writing – review and editing. Overall, each
author made significant revisions to the work and formed the final manuscript draft. Funding
No funding was received for this work. Intellectual property
We confirm that we have given due consideration to the protection of intellectual property associated with this work and that there
are no impediments to publication, including the timing of publication, with respect to intellectual property. In so doing we confirm
that we have followed the regulations of our institutions concerning intellectual property. 8 B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
Research ethics on plant materials
Safflower seeds were used in this study. Both the Turkan and local safflower seeds were kindly provided by Highland oil crops
research department of Holetta Agriculture Research Center, Holeta, Ethiopia. All experimental activities were carried out following
the guidelines provided by Haramaya University and Ethiopian Biodiversity institute.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to
influence the work reported in this paper. Acknowledgements
We are grateful for the assistance provided by the Debre Zeit Agricultural Research Centre, including the use of the laboratory space
and facilities, in all parts of the research and the Biology Department of Haramaya University, School of Graduate Studies. References
[1] F. Akter, S. Wu, M.S. Islam, H. Kyaw, J. Yang, M. Li, Y. Fu, J. Wu, An efficient agrobacterium-mediated genetic transformation system for gene editing in
strawberry (Fragaria× ananassa), Plants 13 (5) (2024) 563.
[2] M. Alam, M. Haque, M. Hossain, S. Sarker, R. Afroz, Haploid plantlet regeneration through anther culture in oilseed Brassica species, Bangladesh J. Agril. Res. 34 (4) (2009) 693–703.
[3] J. Ali, L.C.N. Katrina, A. Shahana, T. Azerkhsh, A.E. Ali, M.M.-N. Corinne, M. Anumalla, Improved anther culture media for enhanced callus formation and plant
regeneration in rice (Oryza sativa L.), Plants 10 (5) (2021) 839.
[4] H. Ali, Ephrem, I. Subas¸ı, M.A. Nawaz, H.J. Chaudhary, M.Q. Shahid, S. Ercis¸li, M.A.B Zia, Mobile genomic element diversity in world collection of safflower
(Carthamus tinctorius L.) panel using iPBS-retrotransposon markers, PLoS One 14 (2) (2019) e0211985.
[5] D. Alimi, A. Hajri, S. Jallouli, H. Sebai, Efficacy of synergistic activity of seed oils from Carthamus tinctorius (Safflower) and Nasturtium officinale (Watercress)
on the lethality of the cattle tick Hyalomma scupense (Acari: ixodidae), Open Vet. J. 12 (1) (2022) 80–90. -80–90.
[6] I. Amghar, M. Ibriz, M. Ibrahimi, A. Boudra, F. Gaboun, R. Meziani, D. Iraqi, M.A. Mazri, G. Diria, R. Abdelwahd, In vitro root induction from argan (Argania
spinosa (L.) Skeels) adventitious shoots: influence of ammonium nitrate, auxins, silver nitrate and putrescine, and evaluation of plantlet acclimatization, Plants 10 (6) (2021) 1062.
[7] S. Belide, L. Hac, S.P. Singh, A.G. Green, C.C. Wood, Agrobacterium-mediated transformation of safflower and the efficient recovery of transgenic plants via
grafting, Plant Methods 7 (2011) 1–13.
[8] J.E. Bowers, S.A. Pearl, J.M. Burke, Genetic mapping of millions of SNPs in safflower (Carthamus tinctorius L.) via whole-genome resequencing, G3: Genes,
Genomes, Genetics 6 (7) (2016) 2203–2211.
[9] N. Burbulis, R. Kupriene, L. Zilenaite, Embryogenesis, callogenesis and plant regeneration from anther cultures of spring rape (Brassica napus L.). Acta
Universitatis Latviensis, Biology (Basel) 676 (2004) 153–158.
[10] R. Cappelletti, S. Sabbadini, B. Mezzetti, The use of TDZ for the efficient in vitro regeneration and organogenesis of strawberry and blueberry cultivars, Sci. Hortic. 207 (2016) 117–124.
[11] Y. Chen, M. Li, J. Wen, X. Pan, Z. Deng, J. Chen, G. Chen, L. Yu, Y. Tang, G. Li, Pharmacological activities of safflower yellow and its clinical applications, Evid.-
Based Complement. Altern. Med. 2022 (1) (2022) 2108557.
[12] H. Cheng, C. Yang, P. Ge, Y. Liu, M.M. Zafar, B. Hu, T. Zhang, Z. Luo, S. Lu, Q. Zhou, Genetic diversity, clinical uses, and phytochemical and pharmacological
properties of safflower (Carthamus tinctorius L.): an important medicinal plant, Front. Pharmacol. 15 (2024) 1374680.
[13] Chung, H., & Ouyang, H. (2020). Use of Thidiazuron for High-Frequency Callus Induction and Organogenesis of Wild Strawberry (Fragaria vesca). Plants 2021,
10, 67: s Note: MDPI stays neu-tral with regard to jurisdictional clai-ms in ….
[14] B. Dash, S.S. Bhuyan, S.K. Singh, M. Chandravani, N. Swain, P. Rout, J.L. Katara, D. BN, S. Samantaray, Androgenesis in indica rice: a comparative competency
in development of doubled haploids, PLoS One 17 (5) (2022) e0267442.
[15] E.T. Dinani, M.R. Shukla, C.E. Turi, J. Sullivan, P.K. Saxena, Thidiazuron: modulator of morphogenesis in vitro. Thidiazuron: from Urea Derivative to Plant
Growth Regulator, 2018, pp. 1–36.
[16] S. Duan, R. Xin, S. Guan, X. Li, R. Fei, W. Cheng, Q. Pan, X. Sun, Optimization of callus induction and proliferation of Paeonia lactiflora Pall. and Agrobacterium-
mediated genetic transformation, Front. Plant Sci. 13 (2022) 996690.
[17] N.C. Ellstrand, H.C. Prentice, J.F. Hancock, Gene Flow and Introgression from Domesticated Plants Into Their Wild Relatives Horizontal gene Transfer, Elsevier, 2002, pp. 217–236.
[18] Emongor, V., & Oagile, O. (2017). Safflower production: botswana University of Agriculture and Natural ResourcesI Gaborone.
[19] A. Flemmer, M. Franchini, L. Lindstr¨om, Description of safflower (Carthamus tinctorius) phenological growth stages according to the extended BBCH scale, Ann.
Appl. Biol. 166 (2) (2015) 331–339.
[20] M.A. Ibrahim, R.A. Ali, E.J. Aldabbagh, A. Mohammed, Factors affecting callus induction from anther and ovary of okra (Abelmoschus esculentus L.), Indian J.
Agril. Res. 57 (5) (2023) 658–664.
[21] M. Javad, A. Zebarjadi, D. Kahrizi, A.H. Salmanian, In vitro propagation and agrobacterium-mediated transformation of safflower (’Carthamus tinctorius’ L.)
using a bacterial mutated aroa gene, Aust. J. Crop Sci. 5 (4) (2011) 479–486.
[22] C. Jing, L. Liu, Z. Wang, Y. Zhang, H. Sun, S. Song, Z. Bai, Z. Lu, C. Li, Nitrogen fertilization increases root growth and coordinates the root–shoot relationship in
cotton, Front. Plant Sci. (2020) 880.
[23] S. Joshi, E. Thoday-Kennedy, H.D. Daetwyler, M. Hayden, G. Spangenberg, S. Kant, High-throughput phenotyping to dissect genotypic differences in safflower
for drought tolerance, PLoS One 16 (7) (2021) e0254908.
[24] N. Khalid, R.S. Khan, M.I. Hussain, M. Farooq, A. Ahmad, I. Ahmed, A comprehensive characterisation of safflower oil for its potential applications as a bioactive
food ingredient-A review, Trends Food Sci. Technol. 66 (2017) 176–186.
[25] Kumar, K.R., Singh, K.P., Jain, P., Raju, D., Kumar, P., Bhatia, R., & Panwar, S. (2018). Influence of growth regulators on callus induction and plant regeneration from anthers of Tagetes spp.
[26] Y. Liang, L. Wang, Carthamus tinctorius L.: a natural neuroprotective source for anti-Alzheimer’s disease drugs, J. Ethnopharmacol. 298 (2022) 115656.
[27] P. Lidder, A. Sonnino, Biotechnologies for the management of genetic resources for food and agriculture, Adv. Genet. 78 (2012) 1–167.
[28] F. Lijiao, G. Meili, Progress of safflower (Carthamus tinctorius L.) regeneration through tissue culture, J. Med. Colleges of PLA 28 (5) (2013) 289–301.
[29] D. Mayakaduwa, T. Silva, Haploid induction in indica rice: exploring new opportunities, Plants 12 (17) (2023) 3118.
[30] D. Mayakaduwa, T.D. Silva, Anther culture as a supplementary tool for rice breeding, Rice Crop-Curr. Dev. (2018) 1–16.
[31] J.F. Menegaes, U.R. Nunes, Safflower: importance, use and economical exploitation, Scientia Agrar. Paranaensis (2020) 1–11. 9 B. Anelay Kassa et al.
Scienti c African 26 (2024) e02367
[32] C. Meneses, M. Valdes-Gonzalez, B.B. Garrido-Su´arez, G. Garrido, Systematic review on the anxiolytic and hypnotic effects of flower extracts in in vivo pre-
clinical studies published from 2010 to 2020, Phytother. Res. 37 (5) (2023) 2144–2167.
[33] D.A. Mosoh, A.K. Khandel, S.K. Verma, W.A. Vendrame, Optimizing callus induction and indirect organogenesis in non-dormant corm explants of Gloriosa
superba (L.) via media priming, Frontiers in Horticulture 3 (2024) 1378098.
[34] J. Motamedi, A. Zebarjadi, D. Kahrizi, A.H. Salmanian, In vitro propagation and agrobacterium-mediated transformation of safflower (’Carthamus tinctorius’ L.)
using a bacterial mutated aroa gene, Aust. J. Crop Sci. 5 (4) (2011) 479–486.
[35] M. Movahedi, V. Ghasemi-Omran, S. Torabi, The effect of different concentrations of TDZ and BA on in vitro regeneration of Iranian cannabis (Cannabis sativa)
using cotyledon and epicotyl explants, J. Plant Mol. Breed. 3 (2) (2015) 20–27.
[36] T. Murashige, F. Skoog, A revised medium for rapid growth and bio assays with tobacco tissue cultures, Physiol. Plant. 15 (3) (1962) 473–497.
[37] D.B. Nakas, S. Avcı, M.D. Kaya, Response of Different Safflower Genotypes to Anther Culture, Kahramanmaras¸ Sütçü ˙Imam Üniversitesi Tarım ve Do˘ga Dergisi 26 (2) (2023) 293–298.
[38] Nauman, K., Khan, R., Hussain, M., Muhammad Farooq, M.F., Asif Ahmad, A.A., & Iftikhar Ahmed, I.A. (2017). A comprehensive characterisation of safflower
oil for its potential applications as a bioactive food ingredient-a review.
[39] U. Nazir, Z. Gul, G.M. Shah, N.I. Khan, Interaction effect of auxin and cytokinin on in vitro shoot regeneration and rooting of endangered medicinal plant
valeriana jatamansi jones through tissue culture, Am. J. Plant. Sci. 13 (2) (2022) 223–240.
[40] M. Niazian, M.E. Shariatpanahi, In vitro-based doubled haploid production: recent improvements, Euphytica 216 (5) (2020) 1–21.
[41] T. Nikam, M. Shitole, In vitro culture of safflower L. cv. Bhima: initiation, growth optimization and organogenesis, Plant. Cell. Tissue. Organ. Cult. 55 (1) (1999) 15–22.
[42] B. Obert, B. Dediˇcov´a, A. Hricov´a, A. Pret’ov´a, Flax anther culture: effect of genotype, cold treatment and media, Plant. Cell. Tissue. Organ. Cult. 79 (2) (2004) 233–238.
[43] Pasandi, M., Janmohammadi, M., Abasi, A., & Sabaghnia, N. (2018). Oil characteristics of safflower seeds under different nutrient and moisture management.
[44] M. Patrascoiu, R. Josef, N. Monica, Z. Rudolf, Perspectives of safflower oil as biodiesel source for South Eastern Europe (comparative study: safflower, soybean
and rapeseed), Fuel 111 (2013) 114–119.
[45] R.B. Prasad, M. Khadeer, P. Seeta, S. Anwar, In vitro induction of androgenic haploids in safflower (Carthamus tinctorius L.), Plant. Cell. Rep. 10 (1991) 48–51.
[46] A. Rayan, N.A. Awad, Production of Haploid Plant Regeneration from Anther Cultures of Apple (Malus domestica BORKH), J. Plant Prod. 33 (5) (2008) 3629–3638.
[47] I.G. Ribeiro, T.C.d. Castro, M.G.P. Coelho, N. Albarello, Effects of different factors on friable callus induction and establishment of cell suspension culture of
Hovenia dulcis (Rhamnaceae), Rodrigu´esia 72 (2021) e00102020.
[48] M. Ruyvaran, A. Zamani, A. Mohamadian, M.M. Zarshenas, M.H. Eftekhari, S. Pourahmad, E.F. Abarghooei, A. Akbari, M. Nimrouzi, Safflower (Carthamus
tinctorius L.) oil could improve abdominal obesity, blood pressure, and insulin resistance in patients with metabolic syndrome: a randomized, double-blind,
placebo-controlled clinical trial, J. Ethnopharmacol. 282 (2022) 114590.
[49] A. Sakina, S. Mir, S. Najeeb, S.M. Zargar, F.A. Nehvi, Z.A. Rather, R.K. Salgotra, A.B. Shikari, Improved protocol for efficacious in vitro androgenesis and
development of doubled haploids in temperate japonica rice, PLoS One 15 (11) (2020) e0241292.
[50] S. Sardouei-Nasab, Z. Nemati, G. Mohammadi-Nejad, R. Haghi, F.R. Blattner, Phylogenomic investigation of safflower (Carthamus tinctorius) and related species
using genotyping-by-sequencing (GBS), Sci. Rep. 13 (1) (2023) 6212.
[51] J.M. Seguí-Simarro, Androgenesis in solanaceae, In vitro embryogenesis in higher plants (2016) 209–244.
[52] S.-G. Seo, S.-H. Ryu, Y. Zhou, S.-H. Kim, Development of an efficient protocol for high-frequency regeneration system in Hibiscus syriacus L, J. Plant Biotechnol. 44 (2) (2017) 164–170.
[53] P. Sharmela, N.M. Ganesan, R. Saraswathi, R. Gnanam, C. Chandrasekhar, Optimization of pre-treatment incubation period on callus induction response in
anthers of selected rice genotypes, Agric. Sci. Dig. 43 (5) (2023) 616–621.
[54] M.R. Shukla, K. Piunno, P.K. Saxena, A.M.P. Jones, Improved in vitro rooting in liquid culture using a two piece scaffold system, Eng. Life Sci. 20 (3–4) (2020) 126–132.
[55] Silva, T., & Ratnayake, W. (2009). Another culture potential of indica rice varieties, KURULU THUDA and BG 250.
[56] K. Sri Shilpa, V. Dinesh Kumar, M. Sujatha, Agrobacterium-mediated genetic transformation of safflower (Carthamus tinctorius L.), Plant Cell, Tissue and Organ
Culture (PCTOC) 103 (3) (2010) 387–401.
[57] S. Srivastava, R. Krishna, R.P. Sinha, M. Singh, TDZ-induced plant regeneration in Brassica oleracea L. var. botrytis: effect of antioxidative enzyme activity and
genetic stability in regenerated plantlets, In Vitro Cellular Dev. Biology-Plant 53 (6) (2017) 598–605.
[58] M. Sujatha, G.S. Dutta, Tissue Culture and Genetic Transformation of Safflower (Carthamus Tinctorius L.) Biotechnology of Neglected and Underutilized Crops, Springer, 2013, pp. 297–318.
[59] K. Talat, S. Anwar, High frequency somatic embryogenesis and plantlet regeneration via somatic embryos in safflower (Carthamus tinctorius L.), Biosciences
Biotechnology Research Asia 7 (1) (2010) 239–249.
[60] T. Waki, M. Terashita, N. Fujita, K. Fukuda, M. Kato, T. Negishi, H. Uchida, Y. Aoki, S. Takahashi, T. Nakayama, Identification of the genes coding for carthamin
synthase, peroxidase homologs that catalyze the final enzymatic step of red pigmentation in safflower (Carthamus tinctorius L.), Plant Cell Physiol. 62 (10) (2021) 1528–1541.
[61] H. Yaman, N.K. Sanyürek, Obtaining haploid plants by irradiated pollen culture in oil seed crops, Biotech Studies 32 (1) (2023) 10–16.
[62] G. Yu, Z. Luo, Y. Zhou, L. Zhang, Y. Wu, L. Ding, Y. Shi, Uncovering the pharmacological mechanism of Carthamus tinctorius L. on cardiovascular disease by a
systems pharmacology approach, Biomed. Pharmacother. 117 (2019) 109094.
[63] M. Zargar, T. Zavarykina, S. Voronov, I. Pronina, M. Bayat, The recent development in technologies for attaining doubled haploid plants in vivo, Agriculture 12 (10) (2022) 1595.
[64] F. Zhou, S. Zhou, S. Cui, J. Liu, Y. Yang, R. Wang, Effects of TDZ and IBA on DNA methylation during callus induction in pear (Pyrus ussuriensis Maxim), Fruit Res. 3 (1) (2023) 1–8. 10
Document Outline
- Effects of plant hormones and genotypes on anther culture response of safflower (Carthamus tinctorius L.)
- Introduction
- Materials and methods
- Plant material
- Culture media and treatments
- Anther culture for callus induction
- Shoot regeneration
- Root induction and acclimatization
- Data analysis
- Result and discussion
- Genotypic effects on callus induction
- Shoot regeneration responses
- Root regeneration and acclimatization
- Conclusion
- CRediT authorship contribution statement
- Funding
- Intellectual property
- Research ethics on plant materials
- Declaration of competing interest
- Acknowledgements
- References


