
















Preview text:
VIETNAM NATIONAL UNIVERSITY – HO CHI MINH CITY UNIVERSITY OF TECHNOLOGY
FACULTY OF CHEMICAL ENGINEERING Chapter 2
Interpretation of Batch Reactor Data
FCE – HCMC University of Technology
Chemical Reaction Engineering Rate data analysis
A rate equation characterizes the rate of reaction
Form of rate equation may either be suggested by • theoretical considerations •
simply be the result of an empirical curve-fitting procedure. •
The value of the constants of the equation can only be found by experiment;
predictive methods are inadequate at present.
The determination of the rate equation is usually a two-step procedure:
1. the concentration dependency is found at fixed temperature
2. the temperature dependence of the rate constants is found, yielding the complete rate equation. Rate data analysis
Equipment by which empirical information is obtained can be: batch and flow reactors.
In batch reactor, the extent of reaction at various times, and this can be followed
• the concentration of a given component.
• the change in some physical property of the fluid, such as the electrical
conductivity or refractive index.
• the change in total pressure of a constant-volume system.
• the change in volume of a constant-pressure system.
The experimental batch reactor is usually operated isothermally and at
constant volume because it is easy to interpret the results of such runs.
The flow reactor is used primarily in the study of the kinetics of heterogeneous reactions. Rate data analysis
Equipment by which empirical information is obtained can be: batch and flow reactors.
In batch reactor, the extent of reaction at various times, and this can be followed •
the concentration of a given component. •
the change in some physical property of the fluid, such as the electrical
conductivity or refractive index. •
the change in total pressure of a constant-volume system. •
the change in volume of a constant-pressure system.
The experimental batch reactor is usually operated isothermally and at constant
volume because it is easy to interpret the results of such runs.
The flow reactor is used primarily in the study of the kinetics of heterogeneous reactions.
There are two procedures for analyzing kinetic data: • the integral • the differential methods
In the integral method of analysis, we guess a particular form of rate equation and,
after appropriate integration and mathematical manipulation, predict that the plot of a
certain concentration function versus time. Types of reaction
❖ Single and Multiple Reactions: •
Single reaction: When a single stoichiometric equation and single rate equation
are chosen to represent the progress of the reaction. •
Multiple reactions: When more than one stoichiometric equation is chosen to
represent the observed changes, then more than one kinetic expression is
needed to follow the changing composition of all the reaction components – Series reactions: – Parallel reactions: Types of reaction
❖ Elementary and Nonelementary Reactions:
Elementary reactions: the rate equation corresponds to a stoichiometric equation are called.
Nonelementary reactions: there is no direct correspondence between stoichiometry and rate. Order of Reaction ❖ Order of the reaction: •
powers to which the concentrations are raised the order of the reaction. •
have a fractional value and need not be an integer. •
Whatever measure we use leaves the order unchanged; •
For rate expressions not of the above form, it makes no sense to use the term reaction order. Conversion •
The fraction of any reactant, say A, converted to something else Batch operation Continuous operation Constant volume
Integral Method of Analysis of Data General procedure: •
Puts a particular rate equation to the test by integrating and comparing the
predicted C versus t curve with the experimental C versus t data. •
If the fit is unsatisfactory, another rate equation is guessed and tested. •
The integral method is especially useful for fitting simple reaction types
corresponding to elementary reactions. •
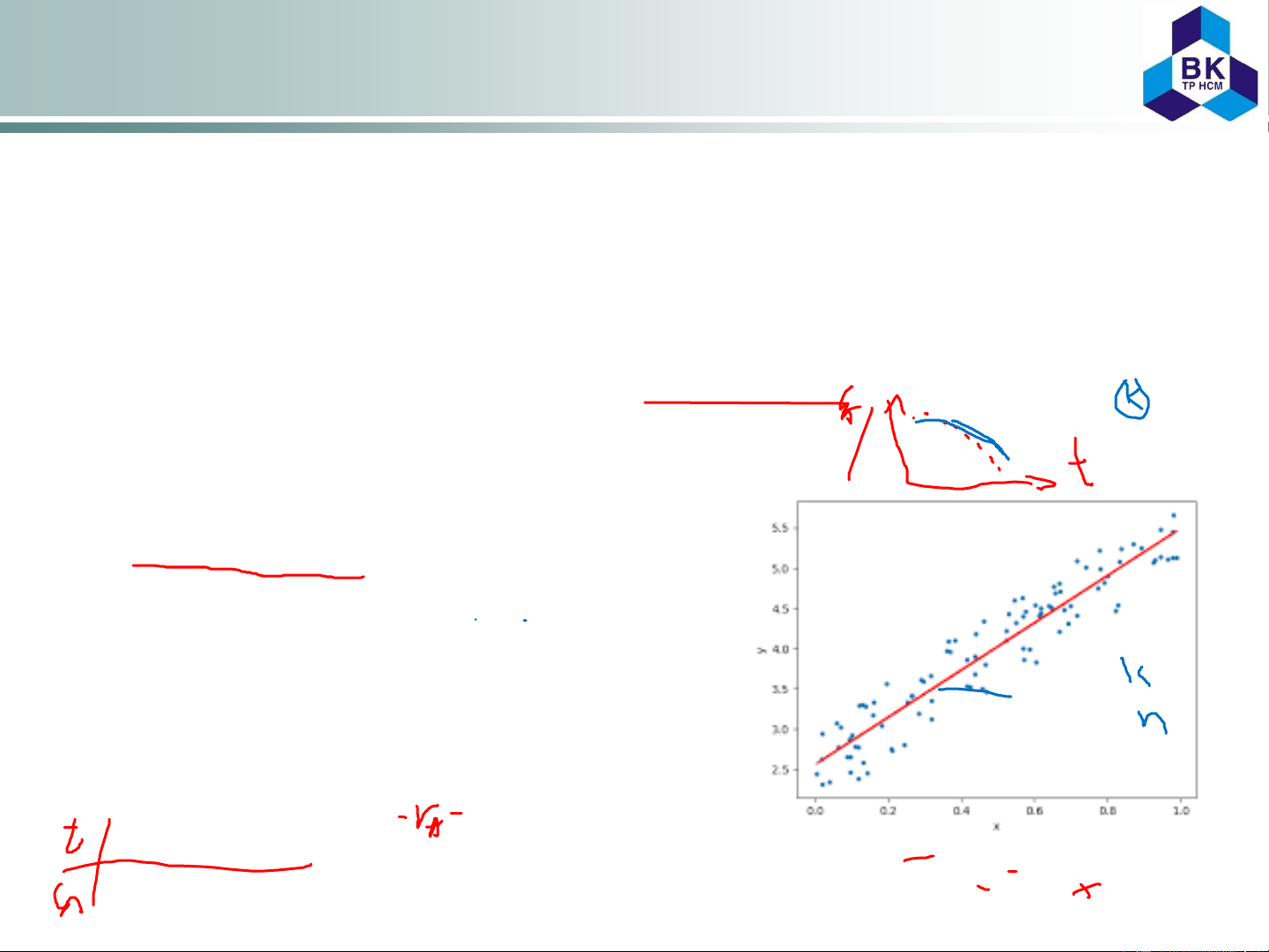
Linear regression is often used
A linear regression line has an
equation of the form Y = a + bX, where
X is the explanatory variable and Y is the dependent variable.
The slope of the line is b, and a is the
intercept (the value of y when x = 0).
Chapter 2. Interpretation of Batch Reactor Data
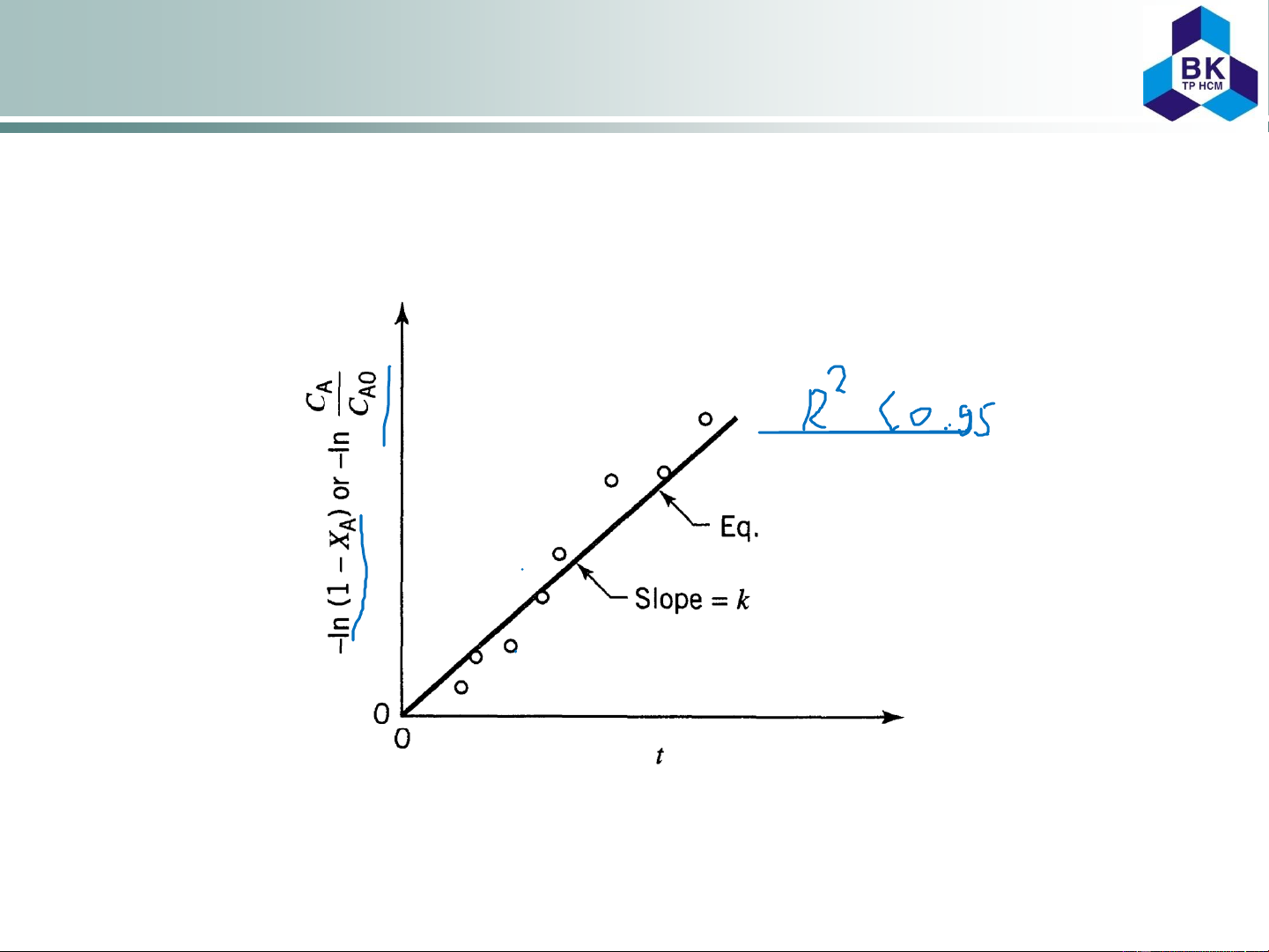
The measured data can be inllustrated in a ln(C /C ) vs. T diagram. The slope A A0
of the straight line leads to the reaction rate constant (Figure 2.3). 2.6 of 2.3
Figure 2.3. Determination of the reaction rate constant k using the
integrated method in a linearised formulation 1
Chapter 2. Interpretation of Batch Reactor Data 1. Rates of reaction
1.1. Description of reaction rates
Reaction rates depend usually in a complex manner on the concentrations, the
temperature and often on the effect introduced by catalysts: r = f ( c ,T, catalyst) i
The order of a reaction is related to the concentration dependence. Typical examples are:
Irreversible Reactions: • First order: A → Products: r = k(T)C k in [s-1] A • Second order: A + B → Products: r = k(T)C C k in [m3 mol-1s-1] A B 2A → Products: r = k(T)C 2 k in [m3 mol-1s-1] A 1
Chapter 2. Interpretation of Batch Reactor Data
Reversible Reactions: • First order: A R: r = k C – k C k in [s-1] 1 A 2 R • Second order: A + B R + S: r = k C C – k C C k in [m3 mol-1s-1] 1 A B 2 R S 2A 2R: r = k C 2 – k C 2 k in [m3 mol-1s-1] 1 A 2 R 2A R + S: r = k C 2 – k C C k in [m3 mol-1s-1] 1 A 2 R S A + B 2R: r = k C C – k C 2 k in [m3 mol-1s-1] 1 A B 2 R
The dependency of the reaction rate on the temperature can be described using Arrhenius law: • E
E is the activation energy of the reaction. A A k T ( ) = k
• k is the pre-exponential factor (not dependent on 0 exp − RT o the reaction temperature 1
Chapter 2. Interpretation of Batch Reactor Data
1.2. Rate laws of simple reactions
In this section, rate equations of simple reactions and the corresponding
temporal change of concentration are analyzed. A closed system (isothermic,
batch reactor) and aconstant volume are assumed.
1.2.1. Irreversible first-order reactions (Decomposition reactions) A → Products 1 dC The rate equation is: i r = = kCi (2.1) dt i dC With i is A and v = -1, then: A r = − = kC (2.2) i A dt CA t dC C The integration form: −
A = k dt A or − ln = (2.3) kt C C C A A0 C Ao 0 A − ln = kt CA0 1
Chapter 2. Interpretation of Batch Reactor Data
The eq. (2.3) leads to the temporal course of concentration c . A −kt C = C e (2.4) A A0 C − C A0 A With X = , the eq. (2.3) leads to: A (2.5) CA0 −kt − 1 ln( − X ) = kt or X = 1− e (2.6) A A
1.2.2. Irreversible bimolecular-type second-order reactions
Consider the reaction (A + B → Products) with corresponding rate equation r = (2.7)
Chapter 2. Interpretation of Batch Reactor Data
Noting that the amounts of A and B that have reacted at any time t are equal and
given by C X , eq. (2.7) can be written in terms of X as A0 A A r = (2.8) (2.9) (2.10)
Chapter 2. Interpretation of Batch Reactor Data
After breakdown into partial fractions, integration, and rearrangement, the final
result in a number of different forms is (2.11)
For the reaction: 2A → Products, the defining second-order differential equation becomes r = (2.12) which on integration yields (2.13)
Chapter 2. Interpretation of Batch Reactor Data
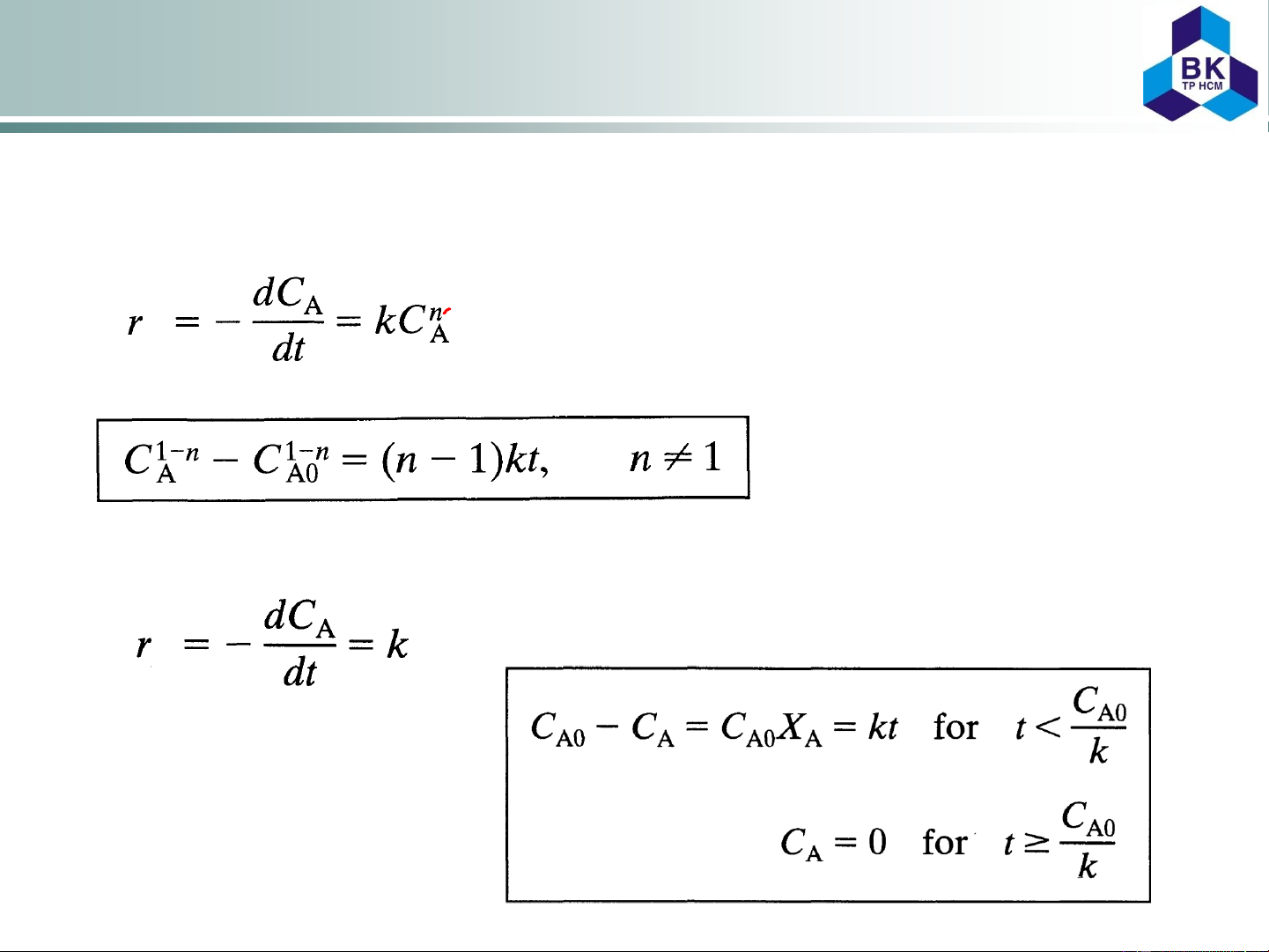
1.2.3. Empirical rate equations of nth order
When the mechanism of reaction is not known, we often attempt to fit the data
with an nth-order rate equation of the form (2.14)
which on separation and integration yields (2.15)
1.2.4. Zero-order reactions
Integrating and noting that CA
can never become negative, (2.16) we obtain directly:
Chapter 2. Interpretation of Batch Reactor Data
1.2.5. Irreversible Reactions in Parallel
Consider the simplest case, A decomposing by two competing paths
The rates of change of the three components are given by (2.17) (2.18) (2.19)
Chapter 2. Interpretation of Batch Reactor Data
The k values are found using all three differential rate equations. First of all,
eq. (2.17), which is of simple first order, is integrated to give (2.20)
Then dividing eq. (2.18) by eq. (2.19) we obtain the following (2.21) which integrated gives simply (2.22)
Chapter 2. Interpretation of Batch Reactor Data
1.2.6. Irreversible Reactions in Series Consider the reaction
Rate eqautions for the three components are (2.23) (2.24) (2.25)

