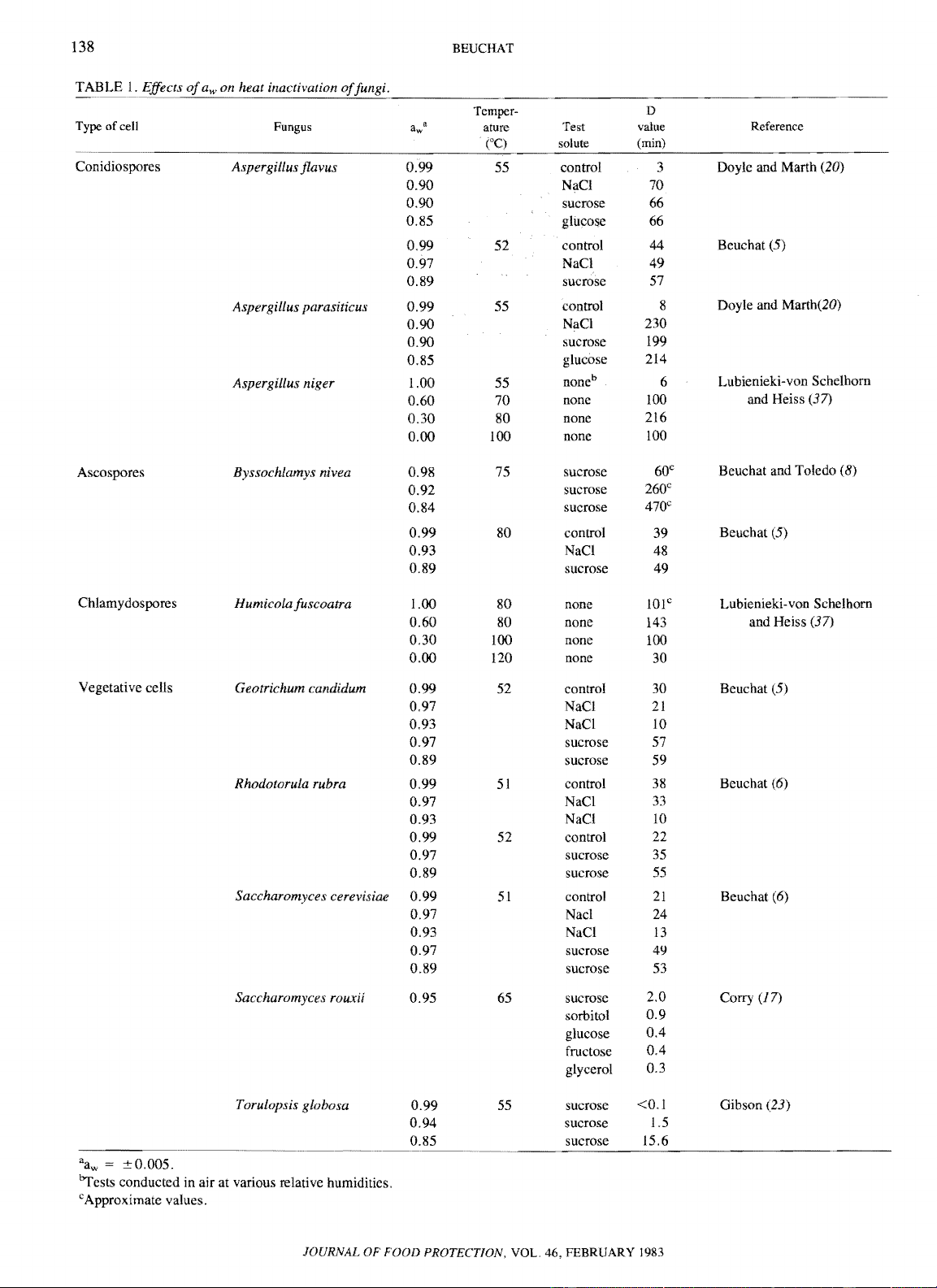
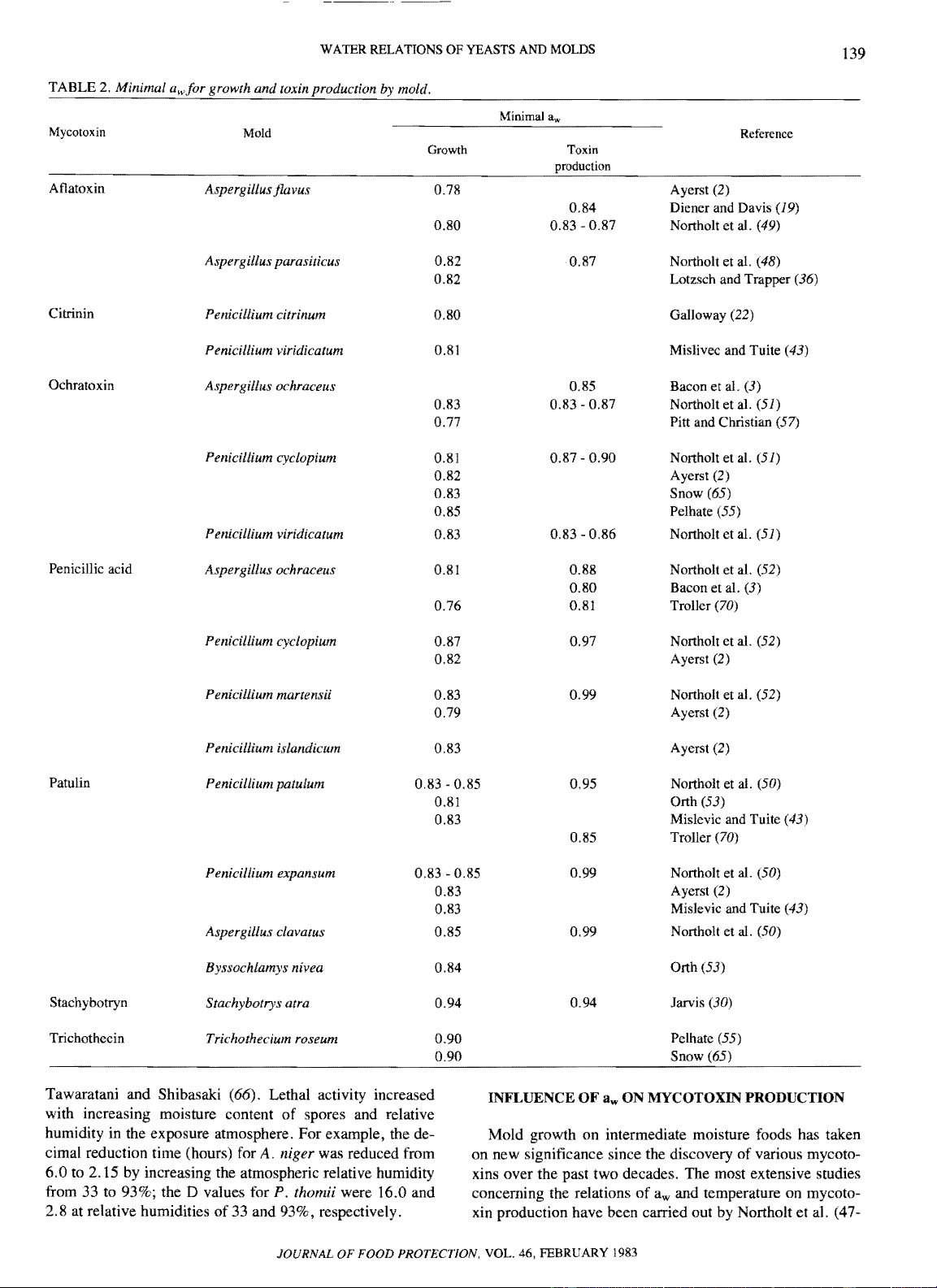
WATER RELATIONS
OF
YEASTS AND MOLDS
141
8. Beuchat,
L.
R.,
and R. T. Toledo. 1977. Behaviour
of
Bys-
sochlamys nivea ascospores in fruit syrups. Trans. Brit. Myco\. Soc.
68:65-71.
9. Bonner,
J. T. 1948. A study
of
the temperature requirements
of
As-
pergillus niger. Mycologia 40:728-738.
10. Brown, A. D. 1974. Microbial water relations: Features
of
the intra-
cellular composition
of
sugar-tolerant yeasts. J. Bacterio\. 118:769-
777.
II.
Brown, A.
D.
1977. Compatible solutes and extreme water stress in
eUkaryotic microorganisms. Adv. Microbial Physio\. 17:181-242.
12. Brown, A.
D.,
and J. R. Simpson. 1972. Water relations
of
sugar-to-
lerant yeasts: the role
of
intracellular polyols. J. Gen. MicrobioL
72:589-591.
13. Charlang, G.
W.,
and N. H. Horowitz. 1971. Germination and
growth
of
Neurospora at low water activities. Proc. Nat!. Acad. Sci.
68:260-262.
14. Charlang, G.
W.,
and N.
H.
Horowitz. 1974. Membrane permeabil-
ity and the loss
of
germination factor from Neurospora crassa at low
water activities.
1.
BacterioL 117:261-264.
15. Clayton, C. N. 1942. The germination
of
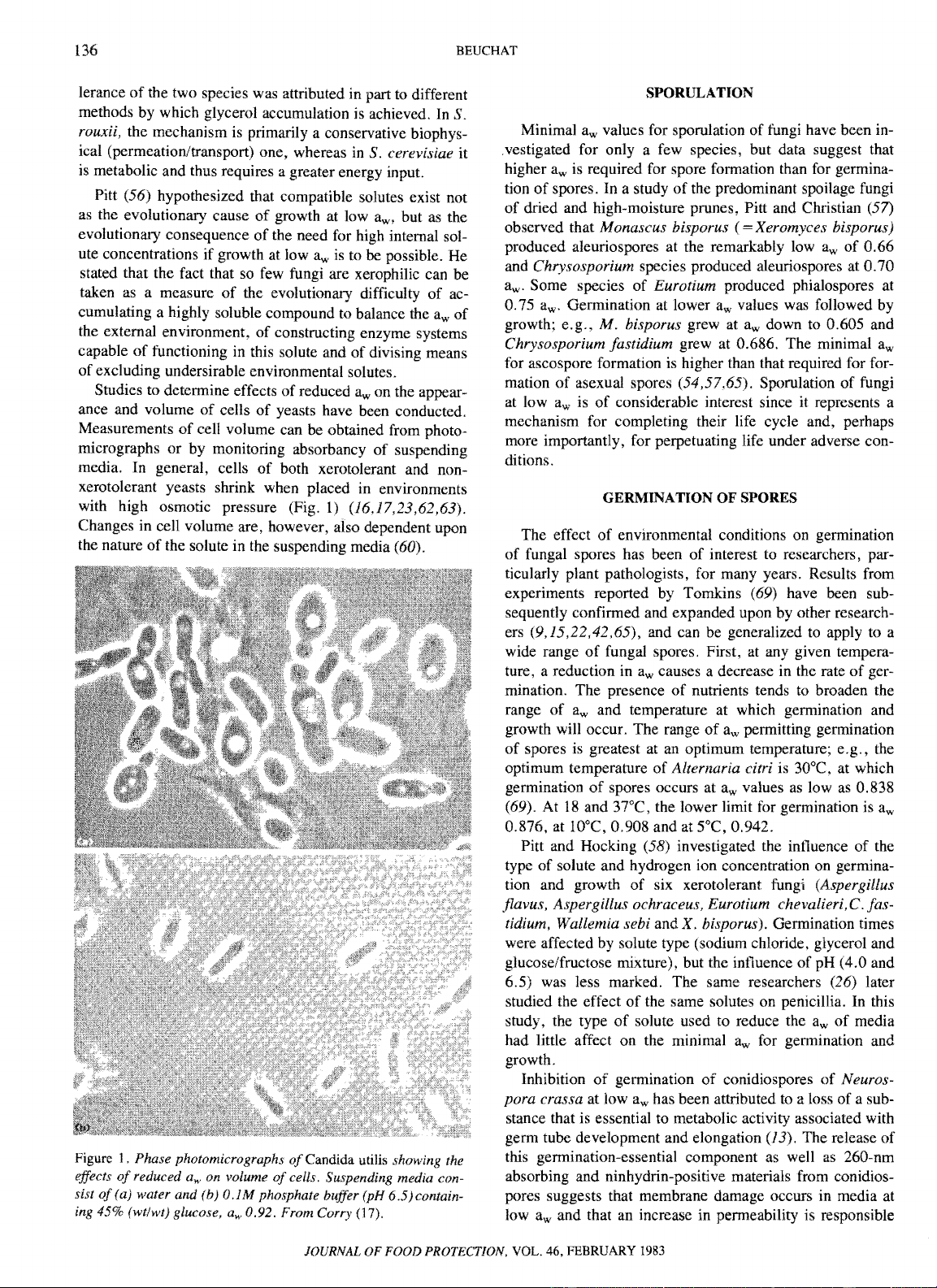
fungus spores in relation to
controlled humidity.
Phytopathology 32:921-943.
16. Corry,
1.
E.
L.
1976. Sugar and polyol permeability
of
Salmonella
and osmophilic yeast cell membranes measured by turbidity, and its
relation to heat resistance. J. App\. Bacteriol. 40:277-284.
17. Corry, J. E.
L. 1976. The effect
of
sugars and polyols on the heat
resistance and morphology
of
osmophilic yeasts.
1.
AppL BacterioL
40:269-276.
18. Corry, J. E.
L.
1978. Relationships
of
water activity to fungal
growth. p. 45-82.
In L. R. Beuchat (ed.), Food and beverage mycol-
ogy. AVI Pub\.
Co.,
Westport, CT.
19. Diener, U.
L.,
and
N.
D. Davis. 1970. Limiting temperature and rel-
ative humidity for aflatoxin production by Aspergillus flavus in stored
peanuts.
J. Am. Oil Chern. Soc. 47:347-351.
20. Doyle,
M.
P.,
and E.
II.
Marth. 1975. Thermal inactivation
of
con-
idia from Aspergillus flavus and Aspergillus parasiticus.
n.
Effects
of
pH
and buffers, glucose, sucrose and sodium chloride. J. Milk
food Technol. 38:750-758.
21. Edgley,
M.,
and A. D. Brown. 1978. Response
of
xerotolerant and
non-tolerant yeasts to water stress.
1.
Gen. Microbio!' 104:343-345.
22. Galloway,
L.
D. 1935. The moisture requirements
of
mold fungi
with special reference to mildew in textiles. J. Textile Inst.
26:Tl23-
129.
23. Gibson,
B.
1973. The effect
of
high sugar concentrations on the heat
resistance
of
vegetative microorganisms. 1. Appl. Bacteriol. 36:365-
376.
24. Gould, G.
W.,
and J.
C.
Measures. 1977. Water relations in single
cells.
Phil. Trans. Royal Soc. London 278:151.
25. Harwig,
J.,
B.
J. Blanchfield, and G. Jarvis. 1977. Effect
of
water
activity on disappearance
of
patulin and citrinin from grains. J. Food
Sci. 42:1225-1228.
26. Hocking, A.
D.,
and J. I. Pitt. 1979. Water relations
of
some
Penicillium species
at
25°C. Trans. Brit. Mycol. Soc. 73:141-145.
27. Hocking, A.
D.,
and J. I. Pitt. 1980. Dichloran-glycerol medium for
enumeration
of
xerophilic fungi from low-moisture foods. Appl. En-
viron. Microbio!. 39:488-492.
28.
Homer,
K. J., and G.
D.
Anagnostopoulos. 1973. Combined effects
of
water activity,
pH
and temperature on the growth and spoilage po-
tential
of
fungi.
1.
AppL Bactcrio!' 36:427-436.
29. Hsieh,
F.-H.,
K. Acot!, and T. P. Labuza. 1975. Prediction
of
microbial death during drying
of
a macaroni product. J. Milk Food
Techno!. 39:619-623.
30. Jarvis, B. 1971. Factors affecting the production
of
mycotoxins. J.
Appl. Bacteriol. 34:199-213.
31. Jarvis,
B.
1978. Methods for detecting fungi in foods and beverages.
p. 471-504. In
L.
R. Beuchat (ed.), Food and beverage mycology.
AVI Publishing
Co.,
Westport, CT.
32. Juven,
B.
J., J. Kanner, and
H.
Weisslowicz. 1978. Influence
of
orange juice composition on the thermal resistance
of
spoilage yeasts.
J. Food Sci. 43:1074-1076,1080.
33. Kushner,
L.,
W.
D.
Rosenweig, and G. Stotzky. 1979. Effects
of
I
saits, sugars, and salt-sugar combinations
on
growth and sporulation
of
an isolate
of
Eurotium rubrum from pancake syrup. J. Food Prot.
41:706-711.
34. Lewis,
D.
H.,
and
D.
C.
Smith. 1967. Sugar alcohols (polyols) in
fungi and green plants.
New
Phytol. 66:143-184.
35. Lloyd, A. C. 1975. Osmophilic yeasts in preserved ginger products.
J. Food Techno!. 10:575-581.
36. Lotzsch,
R.,
and
D.
Trapper. 1978. Aflatoxin and patulin production
as a function
of
water activity (aw-values). Die Fleischwertshaft.
58:2001-2007.
37. Lubieniecki,von Schelhom,
M.,
and R. Heiss. 1975. The influence
of
relative humidity on the thermal resistance
of
mould spores. p.
339-346. In R. B. Duckworth (ed.), Water relations.
of
foods.
Academic Press,
NY.
38. McCrea, A. 1923. Longevity
of
spores
of
Aspergillus oryzae and
Rhizopus nigricans. Science 48:426.
39. Measures,
J. C. 1975. Role
of
amino acids in osmoregulation
of
non-
halophilic bacteria. Nature (London) 257:398-400.
40. Merek, E.
L.,
and
C.
L.
Fergus. 1954. The effect
of
temperature and
relative humidity on the longevity
of
spores
of
oak wilt fungus.
Phytopathology 44:61-64.
41. Miller, M.
W.,
and H. Tanaka. 1963. Microbial spoilage
of
dried
prunes. III. Relation
of
equilibrium relative humidity
to
potential
spoilage. Hilgardia 34:183-190.
42. Mislivec,
P.
B.,
C. T. Dieter, and V. R. Bruce. 1975.
Effectoftem-
perature and relative humidity on spore germination and mycotoxic
species
of
Aspergillus and Penicillium. Mycologia 67: 1187 -1189.
43. Mislivec,
P.
B.,
and J. Tuite. 1970. Temperature and relative humid-
ity requirements
of
species of Penicillium isolated from yellow dent
com
kernels. Mycologia 62:75-88.
44. Munitis, M.
T.,
E. Cabrera, and A. Rodriquez-Navarro. 1976.
An
obligate osmophilic yeast from honey. Appl. Environ. Microbiol.
32:320-323.
45. Murdock, D.
L,
and W.
S.
Hatcher, Jr. 1978. Effect
of
temperature
on survival
of
yeast in 45° and 65° Brix orange concentrate. J. Food
Prot. 41 :689-691 .
46. Northolt, M. D. 1979. The effect
of
water activity and temperature
on the production
of
some mycotoxins. Ph.D. Dissertation.
Rijksinstituut voor de Volksgezondheid, Bilthoven, The Netherlands.
47. Northolt, M.
D.,
and L. B. Bullerman. 1982. Prevention
of
mold
growth and toxin production through control
of
environmental condi-
tions. J. Food Prot. 45:519-526.
48. Northolt, M.
D.,
C.
A. H. Verhulsdonk, P. S. S. Soentoro, and
W.
E. Paulsch. 1976. Effect
of
water activity and temperature on aflato-
xin production by Aspergillus parasiticus. J. Milk Food Techno!.
39:170-174.
49. Northolt, M.
D.,
II.
P. Van Egmond, and W.
E.
Paulsch, 1977. Dif-
ferences betwecn Aspergillus flavus strains in growth and aflatoxin
B
j
production in relation to water activity and temperature.
J.
Food
Prot. 40:778-781.
50. Northolt, M.
D.,
H. P. Van Egmond, and W. E. Paulsch. 1978.
Patulin production by some fungal species in relation to water activ-
ity and temperature. J. Food Prot.
41
:885-890.
51. Northolt, M.
D.,
II.
P. Van Egmond, and W.
E.
Paulsch. 1979. Oc-
hratoxin A production by some fungal species in relation to water ac-
tivity and temperature. J. Food Prot. 42:485-490.
52. Northolt, M. D
..
H. P. Van Egmond, and W. E. Paulsch. 1979.
Penicillic acid production by some fungal species
of
relation to water
activity and temperature. J. Food Prot. 42:476-484.
53.
Orth, R. 1976. The influence
of
water activity on the spore germina-
tion
of
aflatoxin-, sterigmatocystin- and patulin-producing molds.
Lebens. Wiss. Technol.
9:
156-159.
54. Panasenko, V. T. 1967. Ecology
of
microfungi. Bot. Rev. 33:189-
215.
55.
Pelhate,
J.
1968. A study
of
water requirements in some storage
fungi. Mycopathol. Mycol. AppL 36:117-128.
56.
Pitt,
J.
I. 1975. Xerophilic fungi and the spoilage
of
foods
of
plant
origin.
p.
273-307. In R. B. Duckworth (cd.), Water relations
of
foods. Academic Press, London.
57.
Pitt, J., and
H.
Christian. 1968. Water relations in xerophilic fungi
con't. p. 150
JOURNAL OF FOOD PROTECTION, VOL. 46, FEBRUARY 1983
Downloaded from http://meridian.allenpress.com/jfp/article-pdf/46/2/135/1655005/0362-028x-46_2_135.pdf by guest on 13 January 2023